|
This creates a common pathway that is seen with ischemia-reperfusion injuries. This enzyme is released by neutrophils adherent to the damaged endothelial cells of the blood vessels in the brain due to CO toxicity. The cause of the delayed neurologic sequelae is believed to be linked to the lipid peroxidation by toxic oxygen species generated by xanthine oxidase in the brain. The sequelae of CO poisoning can include depression, memory problems, ataxia, behavior problems, and impaired cognitive function. The patient may also develop chest pain, agitation, abdominal pain, drowsiness, loss of consciousness, visual changes, and seizures. 
This results in the classic symptoms of headache, dizziness, nausea, vomiting, flu-like symptoms, fatigue, shortness of breath, impaired judgment, and cognitive dysfunction. This is especially toxic to the heart muscle and the brain. Not only is the oxygen delivery impaired, but 10% to 15% of the CO binds to myoglobin, cytochromes, and NADPH reductase impairing oxidative phosphorylation in the mitochondria of the cells. 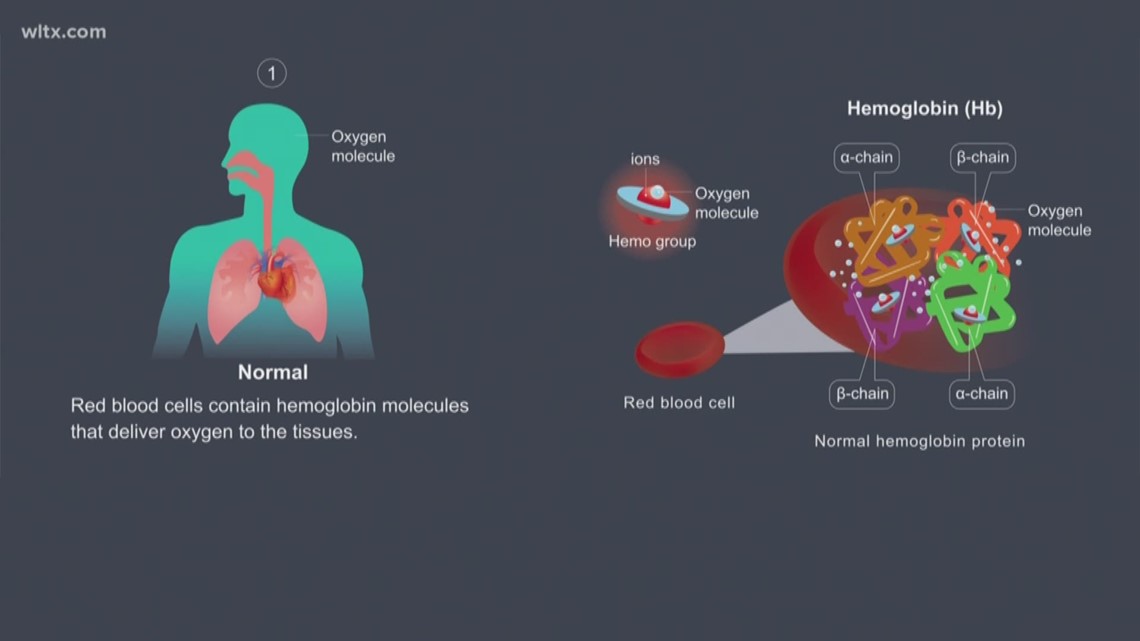
The result of this impairment is decreased oxygen delivery to the tissues and relative hypoxia. The COHb impairs the offloading of the remaining oxygen bound to the hemoglobin molecule (usually, when fully oxygenated the hemoglobin molecule can carry four oxygen molecules). The amount of carboxyhemoglobin formed is dependent on the amount of CO in the environment, duration of exposure, and minute ventilation. This activity reviews the etiology, presentation, evaluation, and management/prevention of CO toxicity and use of hyperbaric oxygen to treat it and reviews the role of the interprofessional team in evaluating, diagnosing, and managing the condition.ĬO is readily absorbed through the lungs and binds to the hemoglobin molecule in red blood cells with an affinity 240 times that of oxygen. Another mechanism of toxicity is from the precipitation of inflammation in the brain resulting in lipid peroxidation and the delayed neurologic sequelae (DNS). 
CO also binds to the cytochrome proteins in mitochondrial impairing cellular respiration. CO binds with a higher affinity than oxygen for the hemoglobin molecule in red blood cells, forming carboxyhemoglobin (COHb), which impairs oxygen offloading resulting in hypoxia in body tissues, especially the brain and heart. This chemical is commonly used in solvents for paint removal and is metabolized to CO in the liver. Poisoning can also occur from inhalation or absorption of methylene chloride (dichloromethane) through the skin. Common sources are engines, non-electric space heaters, furnaces, and smoke from fires. Carbon monoxide (CO) is a toxic gas formed from the incomplete combustion of carbon-containing substances.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
